Latest recommendations

| Id | Title * | Authors * | Abstract * | Picture * | Thematic fields * | Recommender | Reviewers▲ | Submission date | |
|---|---|---|---|---|---|---|---|---|---|
08 Mar 2024
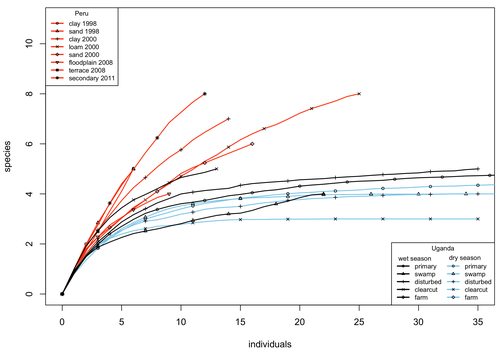
A comparison of the parasitoid wasp species richness of tropical forest sites in Peru and Uganda – subfamily Rhyssinae (Hymenoptera: Ichneumonidae)Tapani Hopkins, Hanna Tuomisto, Isrrael C. Gómez, Ilari E. Sääksjärvi https://doi.org/10.1101/2023.08.23.554460Two sides of tropical richness, parasitoid wasps collected by Malaise traps in tropical rainforests of South America and AfricaRecommended by Giovanny Fagua based on reviews by Mabel Alvarado, Filippo Di Giovanni and 2 anonymous reviewersInsect species richness and diversity comparisons between samples of the tropics around the world are rare, especially in taxa composed mainly of cryptic species as parasitoid wasps. The article by Hopkins et al. (2024) compares samples of parasitoid wasps of the subfamily Rhyssinae (Hymenoptera: Ichneumonidae) collected by Malaise traps in tropical rainforests of Perú and Uganda. The samples presented several differences in the time of collecting, covertures, and the sampling number; however, they used the same kind of traps, and the taxonomic process for species delimitation was made for the same team of ichneumonid experts, using equivalent characters. Publications about this kind of comparative study are difficult to find because cooperative projects on insect richness and diversity from South American and African continents are not frequent. In this sense, this study presented a valuable contrast that shows interesting results about the higher richness and lower abundance of the biota of the American tropics, even with a small sample, in comparison with the biota of the African tropics. The results are supported mainly by the rarefaction curves shown. This pattern of higher species richness and lower specimen abundance, observed in other American tropical taxa such as trees, birds, or butterflies, is observed too in these parasitoid wasps, increasing the body of information that could support the extension of the pattern to the entire biota of the American tropics. The authors recognize the study's limitations, which include strong differences in the size of the forest coverture between places. However, these differences and others are enough described and discussed. This work is useful because it increases the information about the diversity patterns of the tropics around the world and because study a taxon mainly composed of cryptic species, with a small amount of information in tropical regions. References Hopkins T., Tuomisto H., Gómez I.C., Sääksjärvi I. E. 2024. A comparison of the parasitoid wasp species richness of tropical forest sites in Peru and Uganda – subfamily Rhyssinae (Hymenoptera: Ichneumonidae). bioRxiv, ver. 2 peer-reviewed and recommended by Peer Community in Zoology. https://doi.org/10.1101/2023.08.23.554460 | A comparison of the parasitoid wasp species richness of tropical forest sites in Peru and Uganda – subfamily Rhyssinae (Hymenoptera: Ichneumonidae) | Tapani Hopkins, Hanna Tuomisto, Isrrael C. Gómez, Ilari E. Sääksjärvi | <p style="text-align: justify;">The global distribution of parasitoid wasp species richness is poorly known. Past attempts to compare data from different sites have been hampered by small sample sizes and lack of standardisation. During the past d... |  | Biodiversity, Biogeography, Insecta | Giovanny Fagua | 2023-08-24 18:30:26 | View | |
07 Jun 2024

Relationship between weapon size and six key behavioural and physiological traits in males of the European earwigSamantha E.M. Blackwell, Laura Pasquier, Simon Dupont, Séverine Devers, Charlotte Lécureuil, *Joël Meunier https://doi.org/10.1101/2024.03.20.585871The unreliable signal: No correlation between forceps length and male quality in European earwigsRecommended by Olivier Roux based on reviews by Luna Grey and 2 anonymous reviewers based on reviews by Luna Grey and 2 anonymous reviewers
In animals, male weapons such as antlers, horns, spurs, fangs, and tusks typically provide advantages in male contests and increase access to females, thereby enhancing reproductive success. However, such large and extravagant morphological structures are expected to come at a cost, potentially imposing trade-offs with life history traits, physiological functions, or certain behaviors (Emlen, 2001; Emlen, 2008). These costs should be manageable only by males in the best condition. The present study by Blackwell et al. (2024) examines this assumption through a comprehensive study on the European earwig, where males possess forceps-like cerci that vary widely in size within populations. In the European earwig (Forficula auricularia), male forceps are used in male-male contests as weapons to deter competitors prior to mating (Styrsky & Rhein, 1999) or to interrupt mating pairs by non-copulating males (Forslund, 2000; Walker & Fell, 2001). Despite providing benefits in terms of mating success (Eberhard & Gutierrez, 1991; Tomkins & Brown, 2004), it remains unknown whether long or short forceps are associated with other important life-history traits. In this laboratory study, Blackwell et al. (2024) investigated two European earwig populations, each divided into two subpopulations: one with the shortest forceps and one with the longest forceps. They examined the potential costs of long forceps on six different traits: one reproductive trait (sperm storage); three non-reproductive behavioral traits such as locomotor performance (involved in search for resources), fleeing reaction face to a risk (long forceps are supposed to be correlated with boldness), and aggregation behavior (European earwigs are facultative group-living organisms); and survival (when deprived of food and subsequently when exposed to an entomopathogenic fungus). As males in the best condition are supposed to be those that can afford to develop large forceps, Blackwell et al. (2024) predicted that males with long forceps would perform better than those with short forceps across the investigated traits. However, their predictions were not validated, as no correlation between weapon size and male quality was detected in either population. Although the sample size is sometimes limited, the consistency of these results across different populations adds robustness to their conclusions. By demonstrating that forceps length in the European earwig does not reliably indicate male quality, this paper challenges existing theories and highlights the complexity of evolutionary processes shaping morphological traits. Furthermore, the study raises important questions about the evolutionary mechanisms maintaining weapon size diversity, providing a fresh perspective that could stimulate further research and debate in the field, notably the search for other traits where costs might be incurred. References Blackwell, S.E.M., Pasquier, L., Dupont, S., Devers, S., Lécureuil, C. & Meunier, J. (2024). Relationship between weapon size and six key behavioural and physiological traits in males of the European earwig. bioRxiv, ver. 3 peer-reviewed and recommended by Peer Community in Zoology. https://doi.org/10.1101/2024.03.20.585871 Eberhard, W.G., & Gutierrez, E.E. (1991). Male dimorphisms in beetles and earwigs and the question of developmental constraints. Evolution, 45(1), 18–28. https://doi.org/10.2307/2409478 Emlen, D.J. (2001). Costs and the diversification of exaggerated animal structures. Science, 291(5508), 1534–1536. https://doi.org/10.1126/science.1056607 Emlen, D.J. (2008). The evolution of animal weapons. Annual Review of Ecology, Evolution, and Systematics, 39(1), 387–413. https://doi.org/10.1146/annurev.ecolsys.39.110707.173502 Forslund, P. (2000). Male-male competition and large size mating advantage in European earwigs, Forficula auricularia. Animal Behaviour, 59(4), 753–762. https://doi.org/10.1006/anbe.1999.1359 Styrsky, J.D., & Rhein, S.V. (1999). Forceps size does not determine fighting success in European earwigs. Journal of Insect Behavior, 12(4), 475–482. https://doi.org/10.1023/A:1020962606724 Tomkins, J.L., & Brown, G.S. (2004). Population density drives the local evolution of a threshold dimorphism. Nature, 431, 1099–1103. https://doi.org/10.1038/nature02936.1. Walker, K.A., & Fell, R.D. (2001). Courtship roles of male and female European earwigs, Forficula auricularia L. (Dermaptera: Forficulidae), and sexual use of forceps. Journal of Insect Behavior, 14(1), 1–17. https://doi.org/10.1023/A:1007843227591 | Relationship between weapon size and six key behavioural and physiological traits in males of the European earwig | Samantha E.M. Blackwell, Laura Pasquier, Simon Dupont, Séverine Devers, Charlotte Lécureuil, *Joël Meunier | <p style="text-align: justify;">In many animals, male weapons are large and extravagant morphological structures that typically enhance fighting ability and reproductive success. It is generally assumed that growing and carrying large weapons is c... |  | Behavior, Evolution, Insecta, Invertebrates, Life histories, Morphology | Olivier Roux | 2024-03-26 08:56:27 | View | |
19 Jul 2024

Museomics of Carabus giant ground beetles shows an Oligocene origin and in situ Alpine diversificationMarie T. PAULI, Jeremy GAUTHIER, Marjorie LABEDAN, Mickael BLANC, Julia BILAT, Emmanuel F.A. TOUSSAINT https://doi.org/10.1101/2024.03.21.586057Natural history collections continue to inform ground beetle genetics.Recommended by Felix Sperling based on reviews by Michael Caterino, Julian Dupuis and 1 anonymous reviewerSome of the biodiversity of our planet now exists only in museums, due to continuing habitat destruction and climate change. With more than 380 million entomological specimens already preserved in museums (Johnson and Owens 2023), there is much work left to document what we already have. Fortunately, new advances in DNA sequencing have given us the opportunity to get enormous amounts of information from dried specimens on pins. Johnson KR, Owens, (IFP. 2023) A global approach for natural history museum collections. Science 379,1192-1194(2023). https://doi.org/10.1126/science.adf6434 Pauli MT, Gauthier J, Labédan M, Blanc M, Bilat J, Toussaint EFA (2024) Museomics of Carabus giant ground beetles shows an Oligocene origin and in situ alpine diversification. bioRxiv, ver. 5 peer-reviewed and recommended by Peer Community in Zoology. https://doi.org/10.1101/2024.03.21.586057 Schmid, S., Genevest, R., Gobet, E., Suchan, T., Sperisen, C., Tinner, W. and Alvarez, N. (2017), HyRAD-X, a versatile method combining exome capture and RAD sequencing to extract genomic information from ancient DNA. Methods Ecol Evol, 8: 1374-1388. https://doi.org/10.1111/2041-210X.12785 Toussaint EFA, Gauthier J, Bilat J, Gillett CPDT, Gough HM, Lundkvist H, Blanc M, Muñoz-Ramírez CP, Alvarez N (2021) HyRAD-X Exome Capture Museomics Unravels Giant Ground Beetle Evolution, Genome Biology and Evolution, Volume 13, Issue 7, evab112, https://doi.org/10.1093/gbe/evab112 | Museomics of *Carabus* giant ground beetles shows an Oligocene origin and *in situ* Alpine diversification | Marie T. PAULI, Jeremy GAUTHIER, Marjorie LABEDAN, Mickael BLANC, Julia BILAT, Emmanuel F.A. TOUSSAINT | <p style="text-align: justify;">The development of museomics represents a major paradigm shift in the use of natural history collection specimens for systematics and evolutionary biology. New approaches in this field allow the sequencing of hundre... |  | Insecta, Phylogeny, Systematics | Felix Sperling | 2024-03-27 15:30:31 | View | |
01 Jul 2020

Sub-lethal insecticide exposure affects host biting efficiency of Kdr-resistant Anopheles gambiaeMalal Mamadou Diop, Fabrice Chandre, Marie Rossignol, Angelique Porciani, Mathieu Chateau, Nicolas Moiroux, Cedric Pennetier https://doi.org/10.1101/653980kdr homozygous resistant An. gambiae displayed enhanced feeding success when exposed to permethrin Insect-Treated NetsRecommended by Adrian Diaz based on reviews by Thomas Guillemaud, Niels Verhulst, Etienne Bilgo and 1 anonymous reviewerMalaria is a vector-borne parasitic disease found in 91 countries with an estimated of 228 million cases occurred worldwide during 2018. The 93% (213 million) of those cases were reported in the African Region (WHO 2019). Six species of Plasmodium parasites can produce the disease but only P. falciparum and P. vivax are the predominant species globally. More than 40 species of Anopheles mosquitoes are important malaria vectors (Asley et al. 2018). Intrinsic (genetic background, parasite susceptibility) and extrinsic (feeding host preference, host diversity and availability, mosquito abundance) factors affect the capacity of mosquitoes to vector the disease (Macdonald 1952). Malaria is prevented by chemoprophylaxis, vaccination, bite-avoidance and vector-control measures. The mainstays of vector control are long-lasting insecticide (pyrethroid) treated nets and indoor residual spraying with insecticides (Asley et al. 2018). The widespread use of pyrethroid insecticides forced the emergence of insecticide resistance in malaria vectors reducing the insecticidal effect. Mosquitoes can modify their behaviour avoiding insecticide contact and so potentially reducing vector control tools efficacy. In this sense, Diop et al. (2020) investigated whether pre-exposure to an Insecticide-Treated Net (ITN) modulates the mosquito ability to take a blood meal in Anopheles gambiae. By means of video recording experiments the authors analyzed how the feeding/bitting behaviour was affected by kdr mutation genotypes (homozygous susceptible – SS-, heterozygotes -RS- and homozygous resistant -RR-) when exposed to two different insecticides (permethrin and deltamethrin). According to the results, the blood-feeding success did not differ between the three genotypes in the absence of insecticide exposure. However, authors observed differences in the feeding duration and blood meal size. In example, RR mosquitoes spent less time taking their blood meal than RS and SS. On the other hand, RS mosquitoes took higher blood volumes than RR females. These differences can affect the mosquito fitness by decreasing/increasing the likelihood to be killed by the host defensive behavior or increase the oogenesis so enhancing fecundity. Regarding the effect of exposition to insecticides authors detected a strong relationship between kdr genotype and Knock Down (KD) phenotype when mosquitoes were exposed to Permethrin. Previously, the authors have evidenced that RR mosquitoes prefer a host protected by a permethrin-treated net rather than an untreated net and that heterozygotes RS mosquitoes have a remarkable ability to find a hole into a bet net (Diop et al. 2015, Porciani et al. 2017). With data here obtained, they demonstrated that kdr homozygous resistant An. gambiae displayed enhanced feeding success when exposed to permethrin ITN. The changes observed in the feeding/biting mosquito behaviour can affect their fitness shaping the evolution of the insecticide resistance in mosquitoes’ natural populations. Moreover, this may also alter parasite transmission dynamics by modifying vector/host interactions and so vector capacity. References World Health Organization (2019). World malaria report 2019. Geneva: World Health Organization; 2019. ISBN 978-92-4-156572-1 | Sub-lethal insecticide exposure affects host biting efficiency of Kdr-resistant Anopheles gambiae | Malal Mamadou Diop, Fabrice Chandre, Marie Rossignol, Angelique Porciani, Mathieu Chateau, Nicolas Moiroux, Cedric Pennetier | <p>The massive use of insecticide-treated nets (ITNs) has drastically changed the environment for malaria vector mosquitoes, challenging their host-seeking behaviour and biting success. Here, we investigated the effect of a brief exposure to an IT... |  | Behavior, Ecology, Evolution, Medical entomology, Pesticide resistance | Adrian Diaz | Anonymous | 2019-05-29 19:40:25 | View |
21 Mar 2023
Population genetics of Glossina fuscipes fuscipes from southern ChadSophie Ravel, Mahamat Hissène Mahamat, Adeline Ségard, Rafael Argiles-Herrero, Jérémy Bouyer, Jean-Baptiste Rayaisse, Philippe Solano, Brahim Guihini Mollo, Mallaye Pèka, Justin Darnas, Adrien Marie Gaston Belem, Wilfrid Yoni, Camille Noûs, Thierry de Meeûs https://doi.org/10.5281/zenodo.7763870Population genetics of tsetse, the vector of African Trypanosomiasis, helps informing strategies for control programsRecommended by Michael Lattorff based on reviews by 2 anonymous reviewers based on reviews by 2 anonymous reviewers
Human African Trypanosomiasis (HAT), or sleeping sickness, is caused by trypanosome parasites. In sub-Saharan Africa, two forms are present, Trypanosoma brucei gambiense and T. b. rhodesiense, the former responsible for 95% of reported cases. The parasites are transmitted through a vector, Genus Glossina, also known as tsetse, which means fly in Tswana, a language from southern Africa. Through a blood meal, tsetse picks up the parasite from infected humans or animals (in animals, the parasite causes Animal African Trypanosomiasis or nagana disease). Through medical interventions and vector control programs, the burden of the disease has drastically reduced over the past two decades, so the WHO neglected tropical diseases road map targets the interruption of transmission (zero cases) for 2030 (WHO 2022). Meaningful vector control programs utilize traps for the removal of animals and for surveillance, along with different methods of spraying insecticides. However, in existing HAT risk areas, it will be essential to understand the ecology of the vector species to implement control programs in a way that areas cleared from the vector will not be reinvaded from other populations. Thus, it will be crucial to understand basic population genetics parameters related to population structure and subdivision, migration frequency and distances, population sizes, and the potential for sex-biased dispersal. The authors utilize genotyping using nine highly polymorphic microsatellite markers of samples from Chad collected in differently affected regions and at different time points (Ravel et al., 2023). Two major HAT zones exist that are targeted by vector control programs, namely Madoul and Maro, while two other areas, Timbéri and Dokoutou, are free of trypanosomes. Samples were taken before vector control programs started. The sex ratio was female-biased, most strongly in Mandoul and Maro, the zones with the lowest population density. This could be explained by resource limitation, which could be the hosts for a blood meal or the sites for larviposition. Limited resources mean that females must fly further, increasing the chance that more females are caught in traps. The effective population densities of Mandoul and Maro were low. However, there was a convergence of population density and trapping density, which might be explained by the higher preservation of flies in the high-density areas of Timbéri and Dokoutou after the first round of sampling, which can only be tested using a second sampling. The dispersal distances are the highest recorded so far, especially in Mandoul and Maro, with 20-30 km per generation. However, in Timbéri and Dokoutou, which are 50 km apart, very little exchange occurs (approx. 1-2 individuals every six months). A major contributor to this is the massive destruction of habitat that started in the early 1990s and left patchily distributed and fragmented habitats. The Mandoul zone might be safe from reinvasion after eradication, as for a successful re-establishment, either a pair of a female and male or a pregnant female are required. As the trypanosome prevalence amongst humans was 0.02 and of tsetse 0.06 (Ibrahim et al., 2021) before interventions began, medical interventions and vector control might have further reduced these levels, making a reinvasion and subsequent re-establishment of HAT very unlikely. Maro is close to the border of the Central African Republic, and the area has not been well investigated concerning refugee populations of tsetse, which could contribute to a reinvasion of the Maro zone. The higher level of genetic heterogeneity of the Maro population indicates that invasions from neighboring populations are already ongoing. This immigration could also be the reason for not detecting the bottleneck signature in the Maro population. The two HAT areas need different levels of attention while implementing vector eradication programs. While Madoul is relatively safe against reinvasion, Maro needs another type of attention, as frequent and persistent immigration might counteract eradication efforts. Thus, it is recommended that continuous tsetse suppression needs to be implemented in Maro. This study shows nicely that an in-depth knowledge of the processes within and between populations is needed to understand how these populations behave. This can be used to extrapolate, make predictions, and inform the organisations implementing vector control programs to include valuable adjustments, as in the case of Maro. Such integrative approaches can help prevent the failure of programs, potentially saving costs and preventing infections of humans and animals who might die if not treated. References Ibrahim MAM, Weber JS, Ngomtcho SCH, Signaboubo D, Berger P, Hassane HM, Kelm S (2021) Diversity of trypanosomes in humans and cattle in the HAT foci Mandoul and Maro, Southern Chad- Southern Chad-A matter of concern for zoonotic potential? PLoS Neglected Tropical Diseases, 15, e000 323. https://doi.org/10.1371/journal.pntd.0009323 Ravel S, Mahamat MH, Ségard A, Argiles-Herrero R, Bouyer J, Rayaisse JB, Solano P, Mollo BG, Pèka M, Darnas J, Belem AMG, Yoni W, Noûs C, de Meeûs T (2023) Population genetics of Glossina fuscipes fuscipes from southern Chad. Zenodo, ver. 9 peer-reviewed and recommended by PCI Zoology. https://doi.org/10.5281/zenodo.7763870 WHO (2022) Trypanosomiasis, human African (sleeping sickness). https://www.who.int/news-room/fact-sheets/detail/trypanosomiasis-human-african-(sleeping-sickness), retrieved 17. March 2023 | Population genetics of Glossina fuscipes fuscipes from southern Chad | Sophie Ravel, Mahamat Hissène Mahamat, Adeline Ségard, Rafael Argiles-Herrero, Jérémy Bouyer, Jean-Baptiste Rayaisse, Philippe Solano, Brahim Guihini Mollo, Mallaye Pèka, Justin Darnas, Adrien Marie Gaston Belem, Wilfrid Yoni, Camille Noûs, Thierr... | <p>In Subsaharan Africa, tsetse flies (genus Glossina) are vectors of trypanosomes causing Human African Trypanosomiasis (HAT) and Animal African Trypanosomosis (AAT). Some foci of HAT persist in Southern Chad, where a program of tsetse control wa... | Biology, Ecology, Evolution, Genetics/Genomics, Insecta, Medical entomology, Parasitology, Pest management, Veterinary entomology | Michael Lattorff | Audrey Bras | 2022-04-22 11:25:24 | View | |
25 Aug 2021
Up and to the light: intra- and interspecific variability of photo- and geo-tactic oviposition preferences in genus TrichogrammaBurte, V., Perez, G., Ayed, F. , Groussier, G., Mailleret, L., van Oudenhove, L. and Calcagno, V. https://doi.org/10.1101/2021.03.30.437671New insights into oviposition preference of 5 Trichogramma speciesRecommended by Joël Meunier based on reviews by Kévin Tougeron and Eveline C. Verhulst based on reviews by Kévin Tougeron and Eveline C. Verhulst
Insects exhibit a great diversity of life-history traits that often vary not only between species but also between populations of the same species (Flatt and Heyland, 2011). A better understanding of the variation in these traits can be of paramount importance when it comes to species of economic and agricultural interest (Wilby and Thomas, 2002). In particular, the control of the development and expansion of agricultural pests generally requires a good understanding of the parameters that favour the reproduction of these pests and/or the reproduction of the species used to control them (Bianchi et al., 2013; Gäde and Goldsworthy, 2003). Parasitoid wasps of the genus Trichogramma are a classic example of insects involved in pest control (Smith, 1996). This genus comprises over 200 species worldwide, which have been used to control populations of a wide range of lepidopteran pests since the 1900s (Flanders, 1930; Hassan, 1993). Despite its common use, the egg-laying preference of this genus is only partially known. For example, all Trichogramma species are often thought to have positive phototaxis (or negative geotaxis) (e.g. Brower & Cline, 1984; van Atta et al., 2015), but comprehensive studies simultaneously testing this (or other) parameter among Trichogramma species and populations remain rare. This is exactly the aim of the present study (Burte et al., 2021). Using a new experimental approach based on automatic image analysis, the authors compared the photo- and geo-tactic oviposition preference among 5 Trichogramma species from 25 populations. Their results first confirm that most Trichogramma species and populations prefer light to shade, and higher to lower positions for oviposition. Interestingly, they also reveal that the levels of preference for light and gravity show inter- and intraspecific variation (probably due to local adaptation to different strata) and that both preferences tend to relax over time. Overall, this study provides important information for improving the use of Trichogramma species as biological agents. For example, it may help to establish breeding lines adapted to the microhabitat and/or growing parts of plants on which agricultural pests lay eggs most. Similarly, it suggests that the use of multiple strains with different microhabitat selection preferences could lead to better coverage of host plants, as well as a reduction in intraspecific competition in the preferred parts. Finally, this study provides a new methodology to efficiently and automatically study oviposition preferences in Trichogramma, which could be used in other insects with a particularly small size. References Bianchi, F. J. J. A., Schellhorn, N. A. and Cunningham, S. A. (2013). Habitat functionality for the ecosystem service of pest control: reproduction and feeding sites of pests and natural enemies. Agricultural and Forest Entomology, 15, 12–23. https://doi.org/10.1111/j.1461-9563.2012.00586.x Burte V., Perez G., Ayed F., Groussier G., Mailleret L, van Oudenhove L. and Calcagno V. (2021). Up and to the light: intra- and interspecific variability of photo-and geo-tactic oviposition preferences in genus Trichogramma. bioRxiv, 2021.03.30.437671, ver. 4 peer-reviewed and recommended by PCI Zoology. https://doi.org/10.1101/2021.03.30.437671 Brower, J. H. and Cline, L. D. (1984). Response of Trichogramma pretiosum and T. evanescens to Whitelight, Blacklight or NoLight Suction Traps. The Florida Entomologist, 67, 262–268. https://doi.org/10.2307/3493947 Flanders, S. E. (1930). Mass production of egg parasites of the genus Trichogramma. Hilgardia, 4, 465–501. https://doi.org/10.3733/hilg.v04n16p465 Flatt, T. and Heyland, A. (2011). Mechanisms of life history evolution: the genetics and physiology of life history traits and trade-offs. Oxford University Press. https://doi.org/10.1093/acprof:oso/9780199568765.001.0001 Gäde, G. and Goldsworthy, G. J. (2003). Insect peptide hormones: a selective review of their physiology and potential application for pest control. Pest Management Science, 59, 1063–1075. https://doi.org/10.1002/ps.755 Hassan, S. A. (1993). The mass rearing and utilization of Trichogramma to control lepidopterous pests: Achievements and outlook. Pesticide Science, 37, 387–391. https://doi.org/10.1002/ps.2780370412 Smith, S. M. (1996). Biological Control with Trichogramma : Advances, Successes, and Potential of Their Use. Annual Review of Entomology, 41, 375–406. https://doi.org/10.1146/annurev.en.41.010196.002111 van Atta, K. J., Potter, K. A. and Woods, H. A. (2015). Effects of UV-B on Environmental Preference and Egg Parasitization by Trichogramma Wasps (Hymenoptera: Trichogrammatidae). Journal of Entomological Science, 50, 318–325. https://doi.org/10.18474/JES15-09.1 Wilby, A. and Thomas, M. B. (2002). Natural enemy diversity and pest control: patterns of pest emergence with agricultural intensification. Ecology Letters, 5, 353–360. https://doi.org/10.1046/j.1461-0248.2002.00331.x | Up and to the light: intra- and interspecific variability of photo- and geo-tactic oviposition preferences in genus Trichogramma | Burte, V., Perez, G., Ayed, F. , Groussier, G., Mailleret, L., van Oudenhove, L. and Calcagno, V. | <p>Trichogramma are parasitic microwasps much used as biological control agents. The genus is known to harbor tremendous diversity, at both inter- and intra-specific levels. The successful selection of Trichogramma strains for biocontrol depends o... |  | Behavior, Biocontrol, Biodiversity, Ecology, Insecta, Parasitology, Pest management, Systematics, Terrestrial | Joël Meunier | Kévin Tougeron, Eveline C. Verhulst | 2021-04-02 16:10:28 | View |
14 Dec 2023
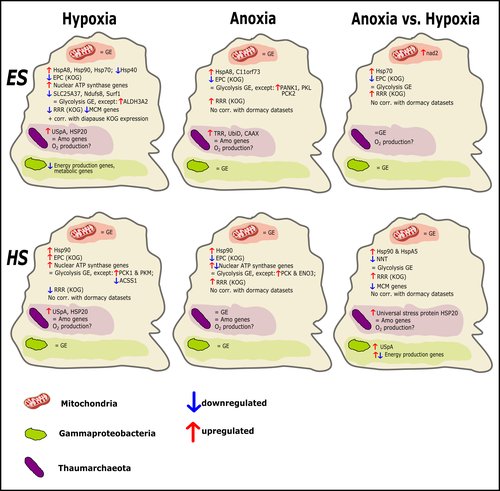
Transcriptomic responses of sponge holobionts to in situ, seasonal anoxia and hypoxiaBrian W Strehlow, Astrid Schuster, Warren R Francis, Lisa Eckford-Soper, Beate Kraft, Rob McAllen, Ronni Nielsen, Susanne Mandrup, Donald E Canfield https://doi.org/10.1101/2023.02.27.530229Future oceanic conditions could leave sponge holobionts breathless – but they won’t let that stop themRecommended by Loïc N. Michel based on reviews by Maria Lopez Acosta and 2 anonymous reviewers based on reviews by Maria Lopez Acosta and 2 anonymous reviewers
It is now widely accepted that anthropogenic climate change is a severe threat to biodiversity, ecosystem function and associated ecosystem services. Assessing the vulnerability of species and predicting their response to future changes has become a priority for environmental biology (Williams et al. 2020). Over the last few decades, oxygen concentrations in both the open ocean and coastal waters have been declining steadily as the result of multiple anthropogenic activities. This global trends towards hypoxia is expected to continue in the future, causing a host of negative effects on marine ecosystems. Oxygen is indeed crucial to many biological processes in the ocean, and its decrease could have strong impacts on biogeochemical cycles, and therefore on marine productivity and biodiversity (Breitburg et al. 2018). Whenever facing such drastic environmental changes, all organisms are expected to have some intrinsic ability to adapt. At shorter than evolutionary timescales, ecological plasticity and the eco-physiological processes that sustain it could constitute important adaptive mechanisms (Williams et al. 2020) Marine sponges seem particularly well-adapted to oxygen deficiency, as some species can survive seasonal anoxia for several months. This paper by Strehlow et al. (2023) examines the mechanisms allowing this exceptional tolerance. Focusing on two species of sponges, they used transcriptomics to assess how gene expression by sponges, by their mitochondria, or by their unique and species-specific microbiome could facilitate this trait. Their results suggest that sponge holobionts maintain metabolic activity under anoxic conditions while displaying shock response, therefore not supporting the hypothesis of sponge dormancy. Furthermore, hypoxia and anoxia seemed to influence gene expression in different ways, highlighting the complexity of sponge response to deoxygenation. As often, their exciting results raise as many questions as they provide answers and pave the way for more research regarding how anoxia tolerance in marine sponges could give them an advantage in future oceanic environmental conditions. References Breitburg et al. (2018): Declining oxygen in the global ocean and coastal waters. Science 359, eaam7240. https://doi.org/10.1126/science.aam7240 Strehlow et al. (2023): Transcriptomic responses of sponge holobionts to in situ, seasonal anoxia and hypoxia. bioRxiv, 2023.02.27.530229, ver. 4 peer-reviewed and recommended by Peer Community in Zoology. https://doi.org/10.1101/2023.02.27.530229 Williams et al. (2008) Towards an Integrated Framework for Assessing the Vulnerability of Species to Climate Change. PLOS Biology 6(12): e325. https://doi.org/10.1371/journal.pbio.0060325 Williams et al. (2020): Research priorities for natural ecosystems in a changing global climate. Global Change Biology 26: 410–416. https://doi.org/10.1111/gcb.14856 | Transcriptomic responses of sponge holobionts to in situ, seasonal anoxia and hypoxia | Brian W Strehlow, Astrid Schuster, Warren R Francis, Lisa Eckford-Soper, Beate Kraft, Rob McAllen, Ronni Nielsen, Susanne Mandrup, Donald E Canfield | <p>Deoxygenation can be fatal for many marine animals; however, some sponge species are tolerant of hypoxia and anoxia. Indeed, two sponge species, <em>Eurypon </em>sp. 2 and <em>Hymeraphia stellifera</em>, survive seasonal anoxia for months at a ... |  | Biology, Ecology, Genetics/Genomics, Invertebrates, Marine, Symbiosis | Loïc N. Michel | Maria Lopez Acosta | 2023-05-12 16:22:47 | View |
27 Apr 2023
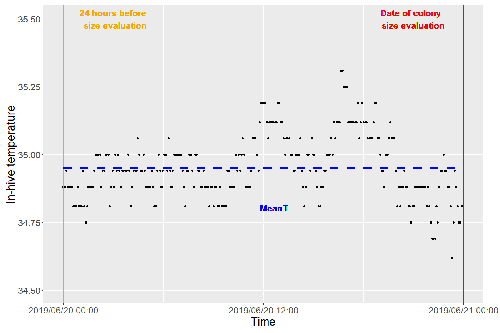
Brood thermoregulation effectivenessis positively linked to the amount of brood but not to the number of bees in honeybee coloniesUgoline Godeau, Maryline Pioz, Olivier Martin, Charlotte Rüger, Didier Crauser, Yves Le Conte, Mickael Henry, Cédric Alaux https://doi.org/10.32942/osf.io/9mwyePrecision and accuracy of honeybee thermoregulationRecommended by Michael Lattorff based on reviews by Jakob Wegener and Christopher Mayack based on reviews by Jakob Wegener and Christopher Mayack
The Western honeybee, Apis mellifera L., is one of the best-studied social insects. It shows a reproductive division of labour, cooperative brood care, and age-related polyethism. Furthermore, honeybees regulate the temperature in the hive. Although bees are invertebrates that are usually ectothermic, this is still true for individual worker bees, but the colony maintains a very narrow range of temperature, especially within the brood nest. This is quite important as the development of individuals is dependent on ambient temperature, with higher temperatures resulting in accelerated development and vice versa. In honeybees, a feedback mechanism couples developmental temperature and the foraging behaviour of the colony and the future population development (Tautz et al., 2003). Bees raised under lower temperatures are more likely to perform in-hive tasks, while bees raised under higher temperatures are better foragers. To maintain optimal levels of worker population growth and foraging rates, it is adaptive to regulate temperature to ensure optimal levels of developing brood. Moreover, this allows honeybees to decouple the internal developmental processes from ambient temperatures enhancing the ecological success of the species. In every system of thermoregulation, whether it is endothermic under the utilization of energetic resources as in mammals or the honeybee or ectothermic as in lower vertebrates and invertebrates through differential exposure to varying environmental temperature gradients, there is a need for precision (low variability) and accuracy (hitting the target temperature). However, in honeybees, the temperature is regulated by workers through muscle contraction and fanning of the wings and thus, a higher number of workers could be better at achieving precise and accurate temperature within the brood nest. Alternatively, the amount of brood could trigger responses with more brood available, a need for more precise and accurate temperature control. The authors aimed at testing these two important factors on the precision and accuracy of within-colony temperature regulation by monitoring 28 colonies equipped with temperature sensors for two years (Godeau et al., 2023). They found that the number of brood cells predicted the mean temperature (accuracy of thermoregulation). Other environmental factors had a small effect. However, the model incorporating these factors was weak in predicting the temperature as it overestimated temperatures in lower ranges and underestimated temperatures in higher ranges. In contrast, the variability of the target temperature (precision of thermoregulation) was positively affected by the external temperature, while all other factors did not show a significant effect. Again, the model was weak in predicting the data. Overall colony size measured in categories of the number of workers and the number of brood cells did not show major differences in variability of the mean temperature, but a slight positive effect for the number of bees on the mean temperature. Unfortunately, the temperature was a poor predictor of colony size. The latter is important as the remote control of beehives using Internet of Things (IoT) technologies get more and more incorporated into beekeeping management. These IoT technologies and their success are dependent on good proxies for the control of the status of the colony. Amongst the factors to monitor, the colony size (number of bees and/or amount of brood) is extremely important, but temperature measurements alone will not allow us to predict colony sizes. Nevertheless, this study showed clearly that the number of brood cells is a crucial factor for the accuracy of thermoregulation in the beehive, while ambient temperature affects the precision of thermoregulation. In the view of climate change, the latter factor seems to be important, as more extreme environmental conditions in the future call for measures of mitigation to ensure the proper functioning of the bee colony, including the maintenance of homeostatic conditions inside of the nest to ensure the delivery of the ecosystem service of pollination. REFERENCES Godeau U, Pioz M, Martin O, Rüger C, Crauser D, Le Conte Y, Henry M, Alaux C (2023) Brood thermoregulation effectiveness is positively linked to the amount of brood but not to the number of bees in honeybee colonies. EcoEvoRxiv, ver. 5 peer-reviewed and recommended by Peer Community in Zoology. https://doi.org/10.32942/osf.io/9mwye Tautz J, Maier S, Claudia Groh C, Wolfgang Rössler W, Brockmann A (2003) Behavioral performance in adult honey bees is influenced by the temperature experienced during their pupal development. PNAS 100: 7343–7347. https://doi.org/10.1073/pnas.1232346100 | Brood thermoregulation effectivenessis positively linked to the amount of brood but not to the number of bees in honeybee colonies | Ugoline Godeau, Maryline Pioz, Olivier Martin, Charlotte Rüger, Didier Crauser, Yves Le Conte, Mickael Henry, Cédric Alaux | <p style="text-align: justify;">To ensure the optimal development of brood, a honeybee colony needs to regulate its temperature within a certain range of values (thermoregulation), regardless of environmental changes in biotic and abiotic factors.... |  | Biology, Conservation biology, Demography/population dynamics, Ecology, Insecta | Michael Lattorff | Mauricio Daniel Beranek | 2022-07-06 09:20:10 | View |
02 Nov 2021

Cuckoo male bumblebees perform slower and longer flower visits than free-living male and worker bumblebeesAlessandro Fisogni, Gherardo Bogo, François Massol, Laura Bortolotti, Marta Galloni https://doi.org/10.5281/zenodo.4489066Cuckoo bumblebee males might reduce plant fitnessRecommended by Michael Lattorff based on reviews by Patrick Lhomme, Silvio Erler and 2 anonymous reviewers based on reviews by Patrick Lhomme, Silvio Erler and 2 anonymous reviewers
In pollinator insects, especially bees, foraging is almost exclusively performed by females due to the close linkage with brood care. They collect pollen as a protein- and lipid-rich food to feed developing larvae in solitary and social species. Bees take carbohydrate-rich nectar in small quantities to fuel their flight and carry the pollen load. To optimise the foraging flight, they tend to be flower constant, reducing the flower handling time and time among individual inflorescences (Goulson, 1999). Males of pollinator species might be found on flowers as well. As they do not collect any pollen for brood care, their foraging flights and visits to flowers might not be shaped by the selective forces that optimise the foraging flights of females. They might stay longer in individual flowers, take up nectar if needed, but might unintentionally carry pollen on their body surface (Wolf & Moritz, 2014). | Cuckoo male bumblebees perform slower and longer flower visits than free-living male and worker bumblebees | Alessandro Fisogni, Gherardo Bogo, François Massol, Laura Bortolotti, Marta Galloni | <p>Cuckoo bumblebees are a monophyletic group within the genus Bombus and social parasites of free-living bumblebees, upon which they rely to rear their offspring. Cuckoo bumblebees lack the worker caste and visit flowers primarily for their own s... |  | Behavior, Biology, Ecology, Insecta, Invertebrates, Terrestrial | Michael Lattorff | Patrick Lhomme, Seth Barribeau , Silvio Erler, Denis Michez | 2021-02-02 01:41:35 | View |
MANAGING BOARD
Dominique Adriaens
Ellen Decaestecker
Benoit Facon
Isabelle Schon
Emmanuel Toussaint
Bertanne Visser










