Latest recommendations

| Id | Title * | Authors * | Abstract * | Picture * | Thematic fields * | Recommender | Reviewers▼ | Submission date | |
|---|---|---|---|---|---|---|---|---|---|
26 Aug 2022
Within and among population differences in cuticular hydrocarbons in the seabird tick Ixodes uriaeMarlène Dupraz, Chloe Leroy, Thorkell Lindberg Thórarinsson, Patrizia d’Ettorre, Karen D. McCoy https://doi.org/10.1101/2022.01.21.477272Seabird tick diversification and cuticular hydrocarbonsRecommended by Felix Sperling based on reviews by 2 anonymous reviewersTicks are notorious vectors of diseases in humans and other vertebrates. Much effort has been expended to understand tick diversity and ecology with the aim of managing their populations to alleviate the misery they bring. Further, the fundamental question of whether ticks are usually host generalists or host specialists has been debated at length and is important both for understanding the mechanisms of their diversification as well as for focusing control of ticks [1]. One elegant resolution of this question is to consider most tick species to be global generalists but local specialists [1]. This is well illustrated in a series of studies of the seabird tick, Ixodes uriae, which is comprised of host-specific races that show genetic [2], morphological [3] and host performance [4] differences associated with the seabirds they feed on. Such a pattern has clear ramifications for sympatric speciation; however, the factors that potentially act to drive these differences have remained elusive. Dupraz et al. [5] have now made intriguing and important steps toward bridging the gap between demonstrating local patterns of tick host association and understanding the physiological mechanisms that may facilitate such divergences. They collected I. uriae ticks from the nests of two seabirds – Atlantic puffins and common guillemots – on the north side of Iceland. Four populations of ticks were sampled, with one island providing both puffin ticks and guillemot ticks, to give two tick populations from each of the two seabird host species. They then washed the ticks in solvent and analyzed the dissolved cuticular hydrocarbons (CHCs) using GC mass spectrometry, revealing 22 different hydrocarbon compounds common to most of these samples. CHCs are known to be important across arthropods for a variety of functions ranging from reducing water loss to facilitating communication and recognition between individuals with species. Dupraz et al. [5] found three hydrocarbons that distinguished puffin ticks most consistently from guillemot ticks. A cross-validation test for host type also assigned 75% of the tick pools to the seabird host of origin. However, with these limited sample sizes, statistical analysis revealed no significant difference in CHC profiles between the host types, although a tendency was evident. Nonetheless, this study revealed a number of potentially diagnostic CHCs for tick host type, as well as some that may be more diagnostic of locations. This provides a fascinating and actionable foundation for further work using additional sites and host types, as well as an entry point into discerning the mechanisms at play in producing the diversity, complexity and adaptability that make ticks such medical menaces. References | Within and among population differences in cuticular hydrocarbons in the seabird tick Ixodes uriae | Marlène Dupraz, Chloe Leroy, Thorkell Lindberg Thórarinsson, Patrizia d’Ettorre, Karen D. McCoy | <p>The hydrophobic layer of the arthropod cuticle acts to maintain water balance, but can also serve to transmit chemical signals via cuticular hydrocarbons (CHC), essential mediators of arthropod behavior. CHC signatures typically vary qualitativ... |  | Acari, Biology, Ecology, Evolution | Felix Sperling | 2022-02-08 13:00:52 | View | |
25 Aug 2022

Improving species conservation plans under IUCN's One Plan Approach using quantitative genetic methodsDrew Sauve, Jane Hudecki, Jessica Steiner, Hazel Wheeler, Colleen Lynch, Amy A. Chabot https://doi.org/10.32942/osf.io/n3zxpQuantitative genetics for a more qualitative conservationRecommended by Peter Galbusera based on reviews by Timothée Bonnet and 1 anonymous reviewerGenetic (bio)diversity is one of three recognised levels of biodiversity, besides species and ecosystem diversity. Its importance for species survival and adaptation is increasingly highlighted and its monitoring recommended (e.g. O’Brien et al 2022). Especially the management of ex-situ populations has a long history of taking into account genetic aspects (through pedigree analysis but increasingly also by applying molecular tools). As in-situ and ex-situ efforts are nowadays often aligned (in a One-Plan-Approach), genetic management is becoming more the standard (supported by quickly developing genomic techniques). However, rarely quantitative genetic aspects are raised in this issue, while its relevance cannot be underestimated. Hence, the current manuscript by Sauve et al (2022) is a welcome contribution, in order to improve conservation efforts. The authors give a clear overview on how quantitative genetic analysis can aid the measurement, monitoring, prediction and management of adaptive genetic variation. The main tools are pedigrees (mainly of ex-situ populations) and the Animal Model. The main goal is to prevent adaption to captivity and altered genetics in general (in reintroduction projects). The confounding factors to take into account (like inbreeding, population structure, differences between facilities, sample size and parental/social effects) are well described by the authors. As such, I fully recommend this manuscript for publication, hoping increased interest in quantitative analysis will benefit the quality of species conservation management. References O'Brien D, Laikre L, Hoban S, Bruford MW et al. (2022) Bringing together approaches to reporting on within species genetic diversity. Journal of Applied Ecology, 00, 1–7. https://doi/10.1111/1365-2664.14225 Sauve D., Spero J., Steiner J., Wheeler H., Lynch C., Chabot A.A. (2022) Improving species conservation plans under IUCN’s One Plan Approach using quantitative genetic methods. EcoEvoRxiv, ver. 9 peer-reviewed and recommended by Peer Community in Zoology. https://doi.org/10.32942/osf.io/n3zxp | Improving species conservation plans under IUCN's One Plan Approach using quantitative genetic methods | Drew Sauve, Jane Hudecki, Jessica Steiner, Hazel Wheeler, Colleen Lynch, Amy A. Chabot | <p>Human activities are resulting in altered environmental conditions that are impacting the demography and evolution of species globally. If we wish to prevent anthropogenic extinction and extirpation, we need to improve our ability to restore wi... |  | Conservation biology, Ecology, Evolution, Genetics/Genomics | Peter Galbusera | 2022-02-21 10:45:22 | View | |
30 Nov 2022
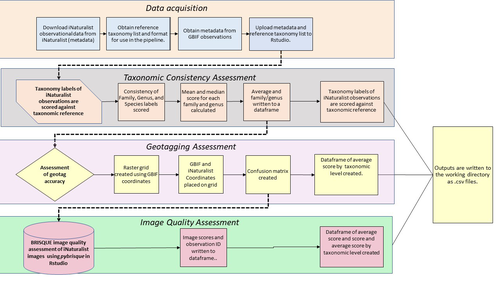
A pipeline for assessing the quality of images and metadata from crowd-sourced databases.Jackie Billotte https://doi.org/10.1101/2022.04.29.490112Harnessing the full potential of iNaturalist and other databasesRecommended by Matthias Foellmer based on reviews by Clive Hambler and Catherine ScottThe popularity of iNaturalist and other online biodiversity databases to which the general public and specialists alike contribute observations has skyrocketed in recent years (Dance 2022). The AI-based algorithms (computer vision) which provide the first identification of a given organism on an uploaded photograph have become very sophisticated, suggesting initial identifications often down to species level with a surprisingly high degree of accuracy. The initial identifications are then confirmed or improved by feedback from the community, which works particularly well for organismal groups to which many active community members contribute, such as the birds. Hence, providing initial observations and identifying observations of others, as well as browsing the recorded biodiversity for given locales or the range of occurrences of individual taxa has become a meaningful and satisfying experience for the interested naturalist. Furthermore, several research studies have now been published relying on observations uploaded to iNaturalist (Szentivanyi and Vincze 2022). However, using the enormous amount of natural history data available on iNaturalist in a systematic way has remained challenging, since this requires not only retrieving numerous observations from the database (in the hundreds or even thousands), but also some level of transparent quality control. Billotte (2022) provides a protocol and R scripts for the quality assessment of downloaded observations from iNaturalist, allowing an efficient and reproducible stepwise approach to prepare a high-quality data set for further analysis. First, observations with their associated metadata are downloaded from iNaturalist, along with the corresponding entries from the Global Biodiversity Information Facility (GBIF). In addition, a taxonomic reference list is obtained (these are available online for many taxa), which is used to assess the taxonomic consistency in the dataset. Second, the geo-tagging is assessed by comparing the iNaturalist and GBIF metadata. Lastly, the image quality is assessed using pyBRISQUE. The approach is illustrated using spiders (Araneae) as an example. Spiders are a very diverse taxon and an excellent taxonomic reference list is available (World Spider Catalogue 2022). However, spiders are not well known to most non-specialists, and it is not easy to take good pictures of spiders without using professional equipment. Therefore, the ability of iNaturalist’s computer vision to provide identifications is limited to this date and the community of specialists active on iNaturalist is comparatively small. Hence, spiders are a good taxon to demonstrate how the pipeline results in a quality-controlled dataset based on crowed-sourced data. Importantly, the software employed is free to use, although inevitably, the initial learning curve to use R scripts can be steep, depending on prior expertise with R/RStudio. Furthermore, the approach is employable with databases other than iNaturalist. In summary, Billotte's (2022) pipeline allows researchers to use the wealth of observations on iNaturalist and other databases to produce large metadata and image datasets of high-quality in a reproducible way. This should pave the way for more studies, which could include, for example, the assessment of range expansions of invasive species or the evaluation of the presence of endangered species, potentially supporting conservation efforts. References Billotte J (2022) A pipeline for assessing the quality of images and metadata from crowd-sourced databases. BiorXiv, 2022.04.29.490112, ver 5 peer reviewed and recommended by Peer Community In Zoology. https://doi.org/10.1101/2022.04.29.490112 Dance A (2022) Community science draws on the power of the crowd. Nature, 609, 641–643. https://doi.org/10.1038/d41586-022-02921-3 Szentivanyi T, Vincze O (2022) Tracking wildlife diseases using community science: an example through toad myiasis. European Journal of Wildlife Research, 68, 74. https://doi.org/10.1007/s10344-022-01623-5 World Spider Catalog (2022). World Spider Catalog. Version 23.5. Natural History Museum Bern, online at http://wsc.nmbe.ch. https://doi.org/10.24436/2 | A pipeline for assessing the quality of images and metadata from crowd-sourced databases. | Jackie Billotte | <p style="text-align: justify;">Crowd-sourced biodiversity databases provide easy access to data and images for ecological education and research. One concern with using publicly sourced databases; however, is the quality of their images, taxonomi... |  | Arachnids, Biodiversity, Biology, Conservation biology, Ecology, Insecta, Invertebrates | Matthias Foellmer | 2022-05-03 00:18:23 | View | |
09 Feb 2023

A novel nematode species from the Siberian permafrost shares adaptive mechanisms for cryptobiotic survival with C. elegans dauer larvaAnastasia Shatilovich, Vamshidhar R. Gade, Martin Pippel, Tarja T. Hoffmeyer, Alexei V. Tchesunov, Lewis Stevens, Sylke Winkler, Graham M. Hughes, Sofia Traikov, Michael Hiller, Elizaveta Rivkina, Philipp H. Schiffer, Eugene W Myers, Teymuras V. Kurzchalia https://doi.org/10.1101/2022.01.28.478251A novel nematode species from the Siberian permafrost shares adaptive mechanisms for cryptobiotic survival with C. elegans dauer larvaRecommended by Isa Schon based on reviews by 3 anonymous reviewersThis article [1] investigated two nematode genera, Panagrolaimus and Plectus, from the Siberian permafrost to unravel the adaptations allowing them to survive cryptobiosis; radio carbon dating showed that the individuals of Panagrolaimus had been in cryobiosis in Siberia for as long as 46,000 years! I was impressed by the multidisciplinary approach of this study, including morphological as well as phylogenetic and -genomic analyses to describe a new species. In triploids as some of the species studied here, it is quite challenging to assemble a novel genome. The authors furthermore not only managed to successfully reanimate the Siberian specimens but could also expose them to repeated freezing and desiccation in the lab, not an easy task. This study reports some amazing discoveries - comparing the molecular toolkits between C. elegans and Panagrolaimus and Plectus revealed that several components were orthologues. Likewise, some of the biochemical mechanisms for surviving freezing in the lab turned out to be similar for C. elegans and the Siberian nematodes. This study thus provides strong evidence that nematodes developed specific mechanisms allowing them to stay in cryobiosis over very long times. A surprising additional experimental result concerns the well-studied C. elegans - dauer larvae of this species can stay viable much longer after periods of animated suspension than previously thought. I highly recommend this article as it is an important contribution to the fields of evolution and molecular biology. This study greatly advanced our understanding of how nematodes could have adapted to cryobiosis. The applied techniques could also be useful for studying similar research questions in other organisms. Reference [1] Shatilovich A, Gade VR, Pippel M, Hoffmeyer TT, Tchesunov AV, Stevens L, Winkler S, Hughes GM, Traikov S, Hiller M, Rivkina E, Schiffer PH, Myers EW, Kurzchalia TV (2023) A novel nematode species from the Siberian permafrost shares adaptive mechanisms for cryptobiotic survival with C. elegans dauer larva. bioRxiv, 2022.01.28.478251, ver. 6 peer-reviewed and recommended by Peer Community in Zoology. https://doi.org/10.1101/2022.01.28.478251 | A novel nematode species from the Siberian permafrost shares adaptive mechanisms for cryptobiotic survival with C. elegans dauer larva | Anastasia Shatilovich, Vamshidhar R. Gade, Martin Pippel, Tarja T. Hoffmeyer, Alexei V. Tchesunov, Lewis Stevens, Sylke Winkler, Graham M. Hughes, Sofia Traikov, Michael Hiller, Elizaveta Rivkina, Philipp H. Schiffer, Eugene W Myers, Teymuras V. K... | <p style="text-align: justify;">Some organisms in nature have developed the ability to enter a state of suspended metabolism called cryptobiosis1 when environmental conditions are unfavorable. This state-transition requires the execution of comple... |  | Ecology, Evolution, Genetics/Genomics | Isa Schon | 2022-05-20 14:32:02 | View | |
21 Jun 2023
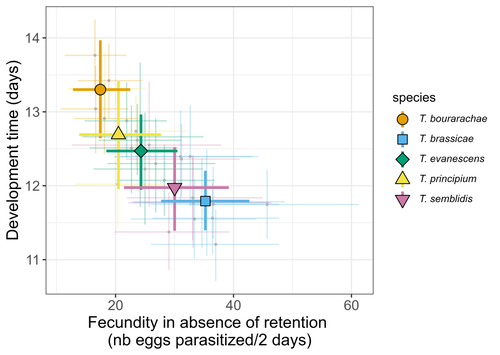
Life-history traits, pace of life and dispersal among and within five species of Trichogramma wasps: a comparative analysisChloé Guicharnaud, Géraldine Groussier, Erwan Beranger, Laurent Lamy, Elodie Vercken, Maxime Dahirel https://doi.org/10.1101/2023.01.24.525360The relationship between dispersal and pace-of-life at different scalesRecommended by Jacques Deere based on reviews by Mélanie Thierry and 1 anonymous reviewerThe sorting of organisms along a fast-slow continuum through correlations between life history traits is a long-standing framework (Stearns 1983) and corresponds to the pace-of-life axis. This axis represents the variation in a continuum of life-history strategies, from fast-reproducing short-lived species to slow-reproducing long-lived species. The pace-of-life axis has been the focus of much research largely in mammals, birds, reptiles and plants but less so in invertebrates (Salguero-Gómez et al. 2016; Araya-Ajoy et al. 2018; Healy et al. 2019; Bakewell et al. 2020). Outcomes from this research have highlighted variation across taxa on this axis and mixed support for, and against, patterns expected of the pace-of-life continuum. Given this, a greater understanding of the variation of the pace-of-life across-, and within, taxa are needed. Indeed, Guicharnard et al. (2023) highlight several points regarding our broader understanding of pace-of-life. In general, invertebrates are poorly represented, the variation of pace-of-life across taxonomic scales is less well understood and the relationship between pace-of-life and dispersal, a key life history, requires more attention. Here, Guicharnard et al. (2023) provide a first attempt at addressing the relationship between dispersal and pace-of-life at different scales. The authors, under controlled conditions, investigated how life-history traits and effective dispersal covary for 28 lines from five species of endoparasitoid wasps from the genus Trichogramma. At the species level negative correlations were found between development time and fecundity, matching pace-of-life axis predictions. Although this correlation was not found to be significant among lines, within species, a similar pattern of a negative correlation was observed. This outcome matches previous findings that consistent pace-of-life axes become more difficult to find at lower taxonomic levels. Unlike the other life-history traits measured, effective dispersal showed no evidence of differences between species or between lines. The authors also found no correlation between effective dispersal and other-life history traits which suggests no dispersal/life-history syndromes in the species investigated. One aspect that was not assessed was the impact of density dependence on pace-of-life and effective dispersal, largely as this was a first step in assessing relationship of dispersal with pace-of-life at different scales. However, the authors do acknowledge the importance of future studies incorporating density dependence and that such studies could potentially lead to more generalizable understanding of pace-of-life and dispersal within Trichogramma. A pleasant addition was the link to potential implications for biocontrol. This addition showed an awareness by the authors of how insights into pace-of-life can have an applied component. The results of the study highlighted that selecting for specific lines of a species, to maximise a trait of interest at the cost of another, may not be as effective as selecting different species when implementing biocontrol. This is especially important as often single, established species used in biocontrol are favoured without consideration of the potential of other species which can lead to more efficient biocontrol. REFERENCES Araya-Ajoy, Y.G., Bolstad, G.H., Brommer, J., Careau, V., Dingemanse, N.J. & Wright, J. (2018). Demographic measures of an individual's "pace of life": fecundity rate, lifespan, generation time, or a composite variable? Behavioral Ecology and Sociobiology, 72, 75. | Life-history traits, pace of life and dispersal among and within five species of *Trichogramma* wasps: a comparative analysis | Chloé Guicharnaud, Géraldine Groussier, Erwan Beranger, Laurent Lamy, Elodie Vercken, Maxime Dahirel | <p>Major traits defining the life history of organisms are often not independent from each other, with most of their variation aligning along key axes such as the pace-of-life axis. We can define a pace-of-life axis structuring reproduction and de... |  | Biology, Ecology, Insecta, Invertebrates, Life histories | Jacques Deere | 2023-01-25 18:15:20 | View | |
14 Nov 2023

Time-course of antipredator behavioral changes induced by the helminth Pomphorhynchus laevis in its intermediate host Gammarus pulex: the switch in manipulation according to parasite developmental stage differs between behaviorsThierry Rigaud, Aude Balourdet, Alexandre Bauer https://doi.org/10.1101/2023.04.25.538244Exploring manipulative strategies of a trophically-transmitted parasite across its ontogenyRecommended by Thierry Lefevre based on reviews by Adèle Mennerat and 1 anonymous reviewerThe intricate relationships between parasites and their hosts often involve a choreography of behavioral changes, with parasites manipulating their hosts in a way that enhances - or seemingly enhances – their transmission (Hughes et al., 2012; Moore, 2002; Poulin, 2010). Host manipulation is increasingly acknowledged as a pervasive adaptive transmission strategy employed by parasites, and as such is one of the most remarkable manifestations of the extended phenotype (Dawkins, 1982). In this laboratory study, Rigaud et al. (2023) delved into the time course of antipredator behavioral modifications induced by the acanthocephalan Pomphorhynchus laevis in its amphipod intermediate host Gammarus pulex. This system has a good foundation of prior knowledge (Bakker et al., 2017; Fayard et al., 2020; Perrot-Minnot et al., 2023), nicely drawn upon for the present work. This parasite orchestrates a switch from predation suppression, during the noninfective phase, to predation enhancement upon maturation. Specifically, G. pulex infected with the non-infective acanthella stage of the parasite can exhibit increased refuge use and reduced activity compared to uninfected individuals (Dianne et al., 2011, 2014), leading to decreased predation by trout (Dianne et al., 2011). In contrast, upon reaching the infective cystacanth stage, the parasite can enhance the susceptibility of its host to trout predation (Dianne et al., 2011). The present work aimed to understand the temporal sequence of these behavioral changes across the entire ontogeny of the parasite. The results confirmed the protective role of P. laevis during the acanthella stage, wherein infected amphipods exhibited heightened refuge use. This protective manipulation, however, became significant only later in the parasite's ontogeny, suggesting a delayed investment strategy, possibly influenced by the extended developmental time of P. laevis. The protective component wanes upon reaching the cystacanth stage, transitioning into an exposure strategy, aligning with theoretical predictions and previous empirical work (Dianne et al., 2011; Parker et al., 2009). The switch was behavior-specific. Unlike the protective behavior, a decline in the amphipod activity rate manifested early in the acanthella stage and persisted throughout development, suggesting potential benefits of reduced activity for the parasite across multiple stages. Furthermore, the findings challenge previous assumptions regarding the condition-dependency of manipulation, revealing that the parasite-induced behavioral changes predominantly occurred in the presence of cues signaling potential predators. Finally, while amphipods infected with acanthella stages displayed survival rates comparable to their uninfected counterparts, increased mortality was observed in those infected with cystacanth stages. Understanding the temporal sequence of host behavioral changes is crucial for deciphering whether it is adaptive to the parasite or not. This study stands out for its meticulous examination of multiple behaviors over the entire ontogeny of the parasite highlighting the complexity and condition-dependent nature of manipulation. The protective-then-expose strategy emerges as a dynamic process, finely tuned to the developmental stages of the parasite and the ecological challenges faced by the host. The delayed emergence of protective behaviors suggests a strategic investment by the parasite, with implications for the host's survival and the parasite's transmission success. The differential impact of infection on refuge use and activity rate further emphasizes the need for a multidimensional approach in studying parasitic manipulation (Fayard et al., 2020). This complexity demands further exploration, particularly in deciphering how trophically-transmitted parasites shape the behavioral landscape of their intermediate hosts and its temporal dynamic (Herbison, 2017; Perrot-Minnot & Cézilly, 2013). As we discover the many subtleties of these parasitic manipulations, new avenues of research are unfolding, promising a deeper understanding of the ecology and evolution of host-parasite interactions. References Bakker, T. C. M., Frommen, J. G., & Thünken, T. (2017). Adaptive parasitic manipulation as exemplified by acanthocephalans. Ethology, 123(11), 779–784. https://doi.org/10.1111/eth.12660 Dawkins, R. (1982). The extended phenotype: The long reach of the gene (Reprinted). Oxford University Press. Dianne, L., Perrot-Minnot, M.-J., Bauer, A., Gaillard, M., Léger, E., & Rigaud, T. (2011). Protection first then facilitation: A manipulative parasite modulates the vulnerability to predation of its intermediate host according to its own developmental stage. Evolution, 65(9), 2692–2698. https://doi.org/10.1111/j.1558-5646.2011.01330.x Dianne, L., Perrot-Minnot, M.-J., Bauer, A., Guvenatam, A., & Rigaud, T. (2014). Parasite-induced alteration of plastic response to predation threat: Increased refuge use but lower food intake in Gammarus pulex infected with the acanothocephalan Pomphorhynchus laevis. International Journal for Parasitology, 44(3–4), 211–216. https://doi.org/10.1016/j.ijpara.2013.11.001 Fayard, M., Dechaume‐Moncharmont, F., Wattier, R., & Perrot‐Minnot, M. (2020). Magnitude and direction of parasite‐induced phenotypic alterations: A meta‐analysis in acanthocephalans. Biological Reviews, 95(5), 1233–1251. https://doi.org/10.1111/brv.12606 Herbison, R. E. H. (2017). Lessons in Mind Control: Trends in Research on the Molecular Mechanisms behind Parasite-Host Behavioral Manipulation. Frontiers in Ecology and Evolution, 5, 102. https://doi.org/10.3389/fevo.2017.00102 Hughes, D. P., Brodeur, J., & Thomas, F. (2012). Host manipulation by parasites. Oxford university press. Moore, J. (2002). Parasites and the behavior of animals. Oxford University Press. Parker, G. A., Ball, M. A., Chubb, J. C., Hammerschmidt, K., & Milinski, M. (2009). When should a trophically transmitted parasite manipulate its host? Evolution, 63(2), 448–458. https://doi.org/10.1111/j.1558-5646.2008.00565.x Perrot-Minnot, M.-J., & Cézilly, F. (2013). Investigating candidate neuromodulatory systems underlying parasitic manipulation: Concepts, limitations and prospects. Journal of Experimental Biology, 216(1), 134–141. https://doi.org/10.1242/jeb.074146 Perrot-Minnot, M.-J., Cozzarolo, C.-S., Amin, O., Barčák, D., Bauer, A., Filipović Marijić, V., García-Varela, M., Servando Hernández-Orts, J., Yen Le, T. T., Nachev, M., Orosová, M., Rigaud, T., Šariri, S., Wattier, R., Reyda, F., & Sures, B. (2023). Hooking the scientific community on thorny-headed worms: Interesting and exciting facts, knowledge gaps and perspectives for research directions on Acanthocephala. Parasite, 30, 23. https://doi.org/10.1051/parasite/2023026 Poulin, R. (2010). Parasite Manipulation of Host Behavior. In Advances in the Study of Behavior (Vol. 41, pp. 151–186). Elsevier. https://doi.org/10.1016/S0065-3454(10)41005-0 Rigaud, T., Balourdet, A., & Bauer, A. (2023). Time-course of antipredator behavioral changes induced by the helminth Pomphorhynchus laevis in its intermediate host Gammarus pulex: The switch in manipulation according to parasite developmental stage differs between behaviors. bioRxiv, ver. 6 peer-reviewed and recommended by Peer Community in Zoology. https://doi.org/10.1101/2023.04.25.538244 | Time-course of antipredator behavioral changes induced by the helminth *Pomphorhynchus laevis* in its intermediate host *Gammarus pulex*: the switch in manipulation according to parasite developmental stage differs between behaviors | Thierry Rigaud, Aude Balourdet, Alexandre Bauer | <p style="text-align: justify;">Many trophically transmitted parasites with complex life cycles manipulate their intermediate host antipredatory defenses in ways facilitating their transmission to final host by predation. Some parasites also prote... |  | Aquatic, Behavior, Crustacea, Invertebrates, Parasitology | Thierry Lefevre | 2023-06-20 15:49:32 | View | |
08 Mar 2024
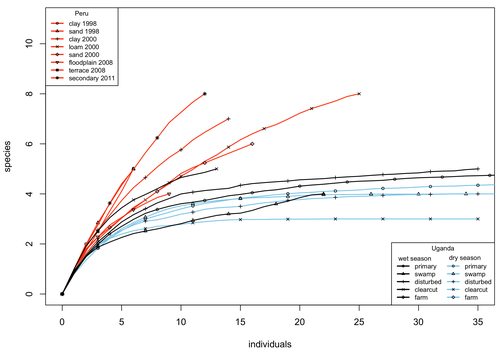
A comparison of the parasitoid wasp species richness of tropical forest sites in Peru and Uganda – subfamily Rhyssinae (Hymenoptera: Ichneumonidae)Tapani Hopkins, Hanna Tuomisto, Isrrael C. Gómez, Ilari E. Sääksjärvi https://doi.org/10.1101/2023.08.23.554460Two sides of tropical richness, parasitoid wasps collected by Malaise traps in tropical rainforests of South America and AfricaRecommended by Giovanny Fagua based on reviews by Mabel Alvarado, Filippo Di Giovanni and 2 anonymous reviewersInsect species richness and diversity comparisons between samples of the tropics around the world are rare, especially in taxa composed mainly of cryptic species as parasitoid wasps. The article by Hopkins et al. (2024) compares samples of parasitoid wasps of the subfamily Rhyssinae (Hymenoptera: Ichneumonidae) collected by Malaise traps in tropical rainforests of Perú and Uganda. The samples presented several differences in the time of collecting, covertures, and the sampling number; however, they used the same kind of traps, and the taxonomic process for species delimitation was made for the same team of ichneumonid experts, using equivalent characters. Publications about this kind of comparative study are difficult to find because cooperative projects on insect richness and diversity from South American and African continents are not frequent. In this sense, this study presented a valuable contrast that shows interesting results about the higher richness and lower abundance of the biota of the American tropics, even with a small sample, in comparison with the biota of the African tropics. The results are supported mainly by the rarefaction curves shown. This pattern of higher species richness and lower specimen abundance, observed in other American tropical taxa such as trees, birds, or butterflies, is observed too in these parasitoid wasps, increasing the body of information that could support the extension of the pattern to the entire biota of the American tropics. The authors recognize the study's limitations, which include strong differences in the size of the forest coverture between places. However, these differences and others are enough described and discussed. This work is useful because it increases the information about the diversity patterns of the tropics around the world and because study a taxon mainly composed of cryptic species, with a small amount of information in tropical regions. References Hopkins T., Tuomisto H., Gómez I.C., Sääksjärvi I. E. 2024. A comparison of the parasitoid wasp species richness of tropical forest sites in Peru and Uganda – subfamily Rhyssinae (Hymenoptera: Ichneumonidae). bioRxiv, ver. 2 peer-reviewed and recommended by Peer Community in Zoology. https://doi.org/10.1101/2023.08.23.554460 | A comparison of the parasitoid wasp species richness of tropical forest sites in Peru and Uganda – subfamily Rhyssinae (Hymenoptera: Ichneumonidae) | Tapani Hopkins, Hanna Tuomisto, Isrrael C. Gómez, Ilari E. Sääksjärvi | <p style="text-align: justify;">The global distribution of parasitoid wasp species richness is poorly known. Past attempts to compare data from different sites have been hampered by small sample sizes and lack of standardisation. During the past d... |  | Biodiversity, Biogeography, Insecta | Giovanny Fagua | 2023-08-24 18:30:26 | View | |
07 Jun 2024

Relationship between weapon size and six key behavioural and physiological traits in males of the European earwigSamantha E.M. Blackwell, Laura Pasquier, Simon Dupont, Séverine Devers, Charlotte Lécureuil, *Joël Meunier https://doi.org/10.1101/2024.03.20.585871The unreliable signal: No correlation between forceps length and male quality in European earwigsRecommended by Olivier Roux based on reviews by Luna Grey and 2 anonymous reviewers based on reviews by Luna Grey and 2 anonymous reviewers
In animals, male weapons such as antlers, horns, spurs, fangs, and tusks typically provide advantages in male contests and increase access to females, thereby enhancing reproductive success. However, such large and extravagant morphological structures are expected to come at a cost, potentially imposing trade-offs with life history traits, physiological functions, or certain behaviors (Emlen, 2001; Emlen, 2008). These costs should be manageable only by males in the best condition. The present study by Blackwell et al. (2024) examines this assumption through a comprehensive study on the European earwig, where males possess forceps-like cerci that vary widely in size within populations. In the European earwig (Forficula auricularia), male forceps are used in male-male contests as weapons to deter competitors prior to mating (Styrsky & Rhein, 1999) or to interrupt mating pairs by non-copulating males (Forslund, 2000; Walker & Fell, 2001). Despite providing benefits in terms of mating success (Eberhard & Gutierrez, 1991; Tomkins & Brown, 2004), it remains unknown whether long or short forceps are associated with other important life-history traits. In this laboratory study, Blackwell et al. (2024) investigated two European earwig populations, each divided into two subpopulations: one with the shortest forceps and one with the longest forceps. They examined the potential costs of long forceps on six different traits: one reproductive trait (sperm storage); three non-reproductive behavioral traits such as locomotor performance (involved in search for resources), fleeing reaction face to a risk (long forceps are supposed to be correlated with boldness), and aggregation behavior (European earwigs are facultative group-living organisms); and survival (when deprived of food and subsequently when exposed to an entomopathogenic fungus). As males in the best condition are supposed to be those that can afford to develop large forceps, Blackwell et al. (2024) predicted that males with long forceps would perform better than those with short forceps across the investigated traits. However, their predictions were not validated, as no correlation between weapon size and male quality was detected in either population. Although the sample size is sometimes limited, the consistency of these results across different populations adds robustness to their conclusions. By demonstrating that forceps length in the European earwig does not reliably indicate male quality, this paper challenges existing theories and highlights the complexity of evolutionary processes shaping morphological traits. Furthermore, the study raises important questions about the evolutionary mechanisms maintaining weapon size diversity, providing a fresh perspective that could stimulate further research and debate in the field, notably the search for other traits where costs might be incurred. References Blackwell, S.E.M., Pasquier, L., Dupont, S., Devers, S., Lécureuil, C. & Meunier, J. (2024). Relationship between weapon size and six key behavioural and physiological traits in males of the European earwig. bioRxiv, ver. 3 peer-reviewed and recommended by Peer Community in Zoology. https://doi.org/10.1101/2024.03.20.585871 Eberhard, W.G., & Gutierrez, E.E. (1991). Male dimorphisms in beetles and earwigs and the question of developmental constraints. Evolution, 45(1), 18–28. https://doi.org/10.2307/2409478 Emlen, D.J. (2001). Costs and the diversification of exaggerated animal structures. Science, 291(5508), 1534–1536. https://doi.org/10.1126/science.1056607 Emlen, D.J. (2008). The evolution of animal weapons. Annual Review of Ecology, Evolution, and Systematics, 39(1), 387–413. https://doi.org/10.1146/annurev.ecolsys.39.110707.173502 Forslund, P. (2000). Male-male competition and large size mating advantage in European earwigs, Forficula auricularia. Animal Behaviour, 59(4), 753–762. https://doi.org/10.1006/anbe.1999.1359 Styrsky, J.D., & Rhein, S.V. (1999). Forceps size does not determine fighting success in European earwigs. Journal of Insect Behavior, 12(4), 475–482. https://doi.org/10.1023/A:1020962606724 Tomkins, J.L., & Brown, G.S. (2004). Population density drives the local evolution of a threshold dimorphism. Nature, 431, 1099–1103. https://doi.org/10.1038/nature02936.1. Walker, K.A., & Fell, R.D. (2001). Courtship roles of male and female European earwigs, Forficula auricularia L. (Dermaptera: Forficulidae), and sexual use of forceps. Journal of Insect Behavior, 14(1), 1–17. https://doi.org/10.1023/A:1007843227591 | Relationship between weapon size and six key behavioural and physiological traits in males of the European earwig | Samantha E.M. Blackwell, Laura Pasquier, Simon Dupont, Séverine Devers, Charlotte Lécureuil, *Joël Meunier | <p style="text-align: justify;">In many animals, male weapons are large and extravagant morphological structures that typically enhance fighting ability and reproductive success. It is generally assumed that growing and carrying large weapons is c... |  | Behavior, Evolution, Insecta, Invertebrates, Life histories, Morphology | Olivier Roux | 2024-03-26 08:56:27 | View | |
19 Jul 2024

Museomics of Carabus giant ground beetles shows an Oligocene origin and in situ Alpine diversificationMarie T. PAULI, Jeremy GAUTHIER, Marjorie LABEDAN, Mickael BLANC, Julia BILAT, Emmanuel F.A. TOUSSAINT https://doi.org/10.1101/2024.03.21.586057Natural history collections continue to inform ground beetle genetics.Recommended by Felix Sperling based on reviews by Michael Caterino, Julian Dupuis and 1 anonymous reviewerSome of the biodiversity of our planet now exists only in museums, due to continuing habitat destruction and climate change. With more than 380 million entomological specimens already preserved in museums (Johnson and Owens 2023), there is much work left to document what we already have. Fortunately, new advances in DNA sequencing have given us the opportunity to get enormous amounts of information from dried specimens on pins. Johnson KR, Owens, (IFP. 2023) A global approach for natural history museum collections. Science 379,1192-1194(2023). https://doi.org/10.1126/science.adf6434 Pauli MT, Gauthier J, Labédan M, Blanc M, Bilat J, Toussaint EFA (2024) Museomics of Carabus giant ground beetles shows an Oligocene origin and in situ alpine diversification. bioRxiv, ver. 5 peer-reviewed and recommended by Peer Community in Zoology. https://doi.org/10.1101/2024.03.21.586057 Schmid, S., Genevest, R., Gobet, E., Suchan, T., Sperisen, C., Tinner, W. and Alvarez, N. (2017), HyRAD-X, a versatile method combining exome capture and RAD sequencing to extract genomic information from ancient DNA. Methods Ecol Evol, 8: 1374-1388. https://doi.org/10.1111/2041-210X.12785 Toussaint EFA, Gauthier J, Bilat J, Gillett CPDT, Gough HM, Lundkvist H, Blanc M, Muñoz-Ramírez CP, Alvarez N (2021) HyRAD-X Exome Capture Museomics Unravels Giant Ground Beetle Evolution, Genome Biology and Evolution, Volume 13, Issue 7, evab112, https://doi.org/10.1093/gbe/evab112 | Museomics of *Carabus* giant ground beetles shows an Oligocene origin and *in situ* Alpine diversification | Marie T. PAULI, Jeremy GAUTHIER, Marjorie LABEDAN, Mickael BLANC, Julia BILAT, Emmanuel F.A. TOUSSAINT | <p style="text-align: justify;">The development of museomics represents a major paradigm shift in the use of natural history collection specimens for systematics and evolutionary biology. New approaches in this field allow the sequencing of hundre... |  | Insecta, Phylogeny, Systematics | Felix Sperling | 2024-03-27 15:30:31 | View |
MANAGING BOARD
Dominique Adriaens
Ellen Decaestecker
Benoit Facon
Isabelle Schon
Emmanuel Toussaint
Bertanne Visser










